Acids Bases and Salts O Level: 5 Proven Facts Every Student Must Know
If there is one topic in O Level Chemistry that shows up consistently across every exam paper, it is acids, bases and salts. Acids bases and salts O level students encounter covers everything from the pH scale and indicators to neutralisation reactions and salt preparation — and it is worth getting right from the start.
In this guide, we will break down the 5 proven facts about acids bases and salts O level students need to know, so you can walk into your chemistry exam with confidence.
You can download the official SEAB O Level Pure Chemistry syllabus (6092) to see exactly how acids, bases and salts fits into the full examination framework alongside other core topics.
What Are Acids, Bases and Salts in O Level Chemistry?
Before we get into the proven facts, it helps to understand the big picture. The acids bases and salts O level topic covers three related families of chemical compounds and how they interact with each other.
- Acids — substances that produce hydrogen ions (H⁺) in aqueous solution. Examples: hydrochloric acid (HCl), sulfuric acid (H₂SO₄), nitric acid (HNO₃)
- Bases — substances that neutralise acids. Bases that dissolve in water are called alkalis and produce hydroxide ions (OH⁻). Examples: sodium hydroxide (NaOH), ammonia solution (NH₃)
- Salts — ionic compounds formed when an acid reacts with a base. The hydrogen in the acid is replaced by a metal ion or ammonium ion. Examples: sodium chloride (NaCl), copper sulfate (CuSO₄)
Understanding how these three compound types relate to each other is the foundation of this entire topic. Once you see the pattern, answering exam questions becomes much more straightforward.
5 Proven Facts About Acids Bases and Salts Every O Level Student Must Know
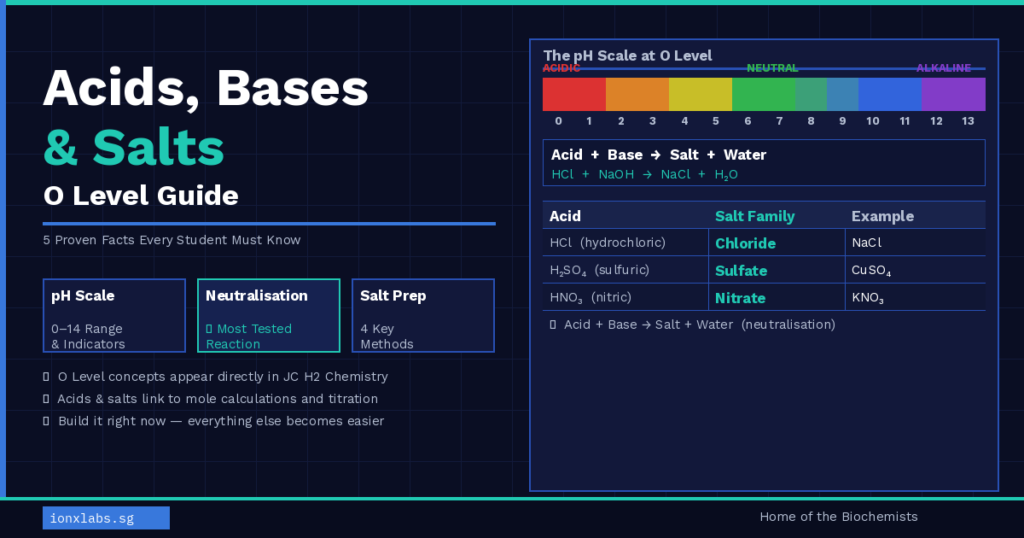
Fact 1: The pH Scale Measures Acidity and Alkalinity
The pH scale runs from 0 to 14 and tells you how acidic or alkaline a solution is:
- pH 0–6 = acidic (lower pH = stronger acid)
- pH 7 = neutral (e.g. pure water)
- pH 8–14 = alkaline (higher pH = stronger alkali)
At O Level, you need to know not just the pH values but also how to use universal indicator and a pH meter to measure pH. A common exam question asks you to state the colour of universal indicator at a given pH — make sure you memorise the colour sequence: red → orange → yellow → green → blue → violet.
It’s also important to understand that pH is a logarithmic scale — a solution of pH 3 is ten times more acidic than a solution of pH 4. This concept sometimes appears in higher-order questions.
Fact 2: Strong Acids and Weak Acids Are Not the Same as Concentrated and Dilute
This is one of the most common points of confusion in the acids bases and salts O level topic — and examiners know it.
- Strong acids (e.g. HCl, H₂SO₄, HNO₃) fully ionise in water — all molecules break up into ions
- Weak acids (e.g. ethanoic acid, carbonic acid) only partially ionise in water
- Concentrated refers to the amount of acid dissolved — a high concentration of molecules
- Dilute refers to a low concentration — fewer molecules per unit volume
You can have a concentrated weak acid or a dilute strong acid — these are independent properties. Examiners regularly test this distinction in Paper 2 structured questions.
Fact 3: Neutralisation Always Produces a Salt and Water
The core reaction in the acids bases and salts O level topic is neutralisation. When an acid reacts with a base, it always produces a salt and water:
Acid + Base → Salt + Water
The name of the salt produced depends on which acid and which base react:
| Acid Used | Salt Family Produced | Example |
|---|---|---|
| Hydrochloric acid (HCl) | Chloride | Sodium chloride (NaCl) |
| Sulfuric acid (H₂SO₄) | Sulfate | Copper sulfate (CuSO₄) |
| Nitric acid (HNO₃) | Nitrate | Potassium nitrate (KNO₃) |
Being able to identify and name salts correctly is a guaranteed source of marks in the O Level Chemistry exam. Practise writing full ionic equations for neutralisation reactions — not just word equations.
Fact 4: There Are Four Main Methods of Salt Preparation
Salt preparation is one of the most practical and heavily tested sections within acids bases and salts. O Level students need to know which method to use depending on whether the salt is soluble or insoluble:
- Titration — used to prepare soluble salts from an acid and an alkali (e.g. NaCl from HCl and NaOH)
- Excess solid method — used to prepare soluble salts from an acid and a metal, metal oxide, or metal carbonate
- Precipitation — used to prepare insoluble salts by mixing two soluble solutions
- Direct combination — used in specific cases like iron sulfide formation
A very common exam question presents a scenario and asks you to select the appropriate method and describe the steps. Make sure you understand why each method is chosen, not just what the steps are.
Fact 5: Indicators Tell You Whether a Solution Is Acidic or Alkaline
Indicators are substances that change colour depending on the pH of a solution. At O Level, you need to know these key indicators:
| Indicator | Colour in Acid | Colour in Neutral | Colour in Alkali |
|---|---|---|---|
| Litmus | Red | Purple | Blue |
| Methyl orange | Red | Orange | Yellow |
| Phenolphthalein | Colourless | Colourless | Pink |
| Universal indicator | Red/Orange/Yellow | Green | Blue/Violet |
Phenolphthalein and methyl orange are particularly important for titration experiments. You need to know which indicator to choose and why — phenolphthalein is preferred for strong acid/strong alkali titrations because it gives a sharp colour change at the endpoint.
How to Study Acids Bases and Salts for O Level Chemistry
The acids bases and salts O level topic is one where a systematic approach pays off enormously. Here’s how to study it effectively:
Master the Naming System First
Before you tackle reactions, make sure you can confidently name any salt given the acid and base. Drill this until it is automatic: HCl → chloride, H₂SO₄ → sulfate, HNO₃ → nitrate. This single skill unlocks a large number of exam marks.
Write Out Equations by Hand
The best way to remember neutralisation reactions is to write them out repeatedly — word equations first, then symbol equations, then full ionic equations. If you can write a balanced ionic equation from memory, you’re operating at distinction level.
Understand Salt Preparation as a Decision Tree
When a question asks how to prepare a salt, ask yourself: is the salt soluble or insoluble? If soluble, what are the starting materials? Walking through this logic — rather than memorising steps — means you can handle any variation the examiner throws at you. For more strategies on approaching O Level Chemistry questions, read our guide on top tips to score well in O Level Chemistry.
Struggling With Acids, Bases and Salts? You Are Not Alone
Many O Level Chemistry students find acids bases and salts challenging because it combines conceptual understanding (pH, ionisation) with practical knowledge (salt preparation methods) and calculation skills (titration). It is a topic where gaps in understanding compound quickly.
If you find yourself confused about why Pure Chemistry feels harder than expected, our post on why Pure Chemistry suddenly feels harder in Sec 4 breaks down exactly what changes and what you can do about it.
At IONX Labs Learning Centre, our O Level Chemistry classes cover acids, bases and salts in depth — building from first principles so students understand the logic behind every reaction, not just the steps. Our tutors use past paper questions and targeted practice to make sure every student can handle whatever the examiner puts in front of them.
You might also find it useful to revisit related topics — our guides on what is organic chemistry and the mole concept tie in closely with acids, bases and salts at the higher levels of the O Level Chemistry paper.
Ready to Master Acids, Bases and Salts for O Level?
Whether you’re just starting out on this topic or trying to close gaps before the exam, IONX Labs is here to help. Our O Level Chemistry classes are small, focused, and built around the Singapore syllabus. Find out more about how our O Level Pure Chemistry tuition programme is structured and what results our students achieve.
📲 WhatsApp us today to find out more or to book your first session.
No pressure — just a conversation about how we can help your child score better in Chemistry.
Frequently Asked Questions
What are acids, bases and salts in O Level Chemistry?
Acids are substances that produce hydrogen ions (H⁺) in solution. Bases neutralise acids, and alkalis are bases that dissolve in water to produce hydroxide ions (OH⁻). Salts are ionic compounds formed when an acid reacts with a base — the hydrogen in the acid is replaced by a metal ion.
What is the difference between a strong acid and a concentrated acid?
These are two separate properties that are often confused. A strong acid fully ionises in water, while a weak acid only partially ionises. Concentration refers to how much acid is dissolved — you can have a dilute strong acid or a concentrated weak acid. This distinction is commonly tested in O Level Chemistry exams.
How do you name a salt in O Level Chemistry?
The name of a salt comes from the acid and base used to make it. Hydrochloric acid produces chloride salts, sulfuric acid produces sulfate salts, and nitric acid produces nitrate salts. The first part of the salt name comes from the metal or ammonium ion in the base.
What are the methods of salt preparation at O Level?
There are four main methods: titration (for soluble salts from acid and alkali), the excess solid method (for soluble salts from acid and metal, metal oxide or carbonate), precipitation (for insoluble salts), and direct combination. Choosing the right method depends on whether the salt is soluble or insoluble.
Which indicators do I need to know for O Level Chemistry?
You need to know litmus (red in acid, blue in alkali), methyl orange (red in acid, yellow in alkali), phenolphthalein (colourless in acid, pink in alkali), and universal indicator (red/orange/yellow in acid, green when neutral, blue/violet in alkali). Phenolphthalein and methyl orange are most important for titration experiments.
